PRESS RELEASE
Contact: Paul Tunnecliff
800-684-4549 phone
registration@hcconferences.com
WASHINGTON DC USA -- HEALTHCARE UPDATE NEWS SERVICETM - NOVEMBER 14, 2003: Today at the Fourth Annual Pharmaceutical Regulatory Compliance Congress and Best Practices Forum sponsored by the Pharmaceutical Compliance Forum the 400 attendees representing the nation's leading pharmaceutical enterprises expressed the need for further governmental guidance regarding regulatory matters.
PHARMACEUTICAL SALES AND MARKETING GUIDANCE:
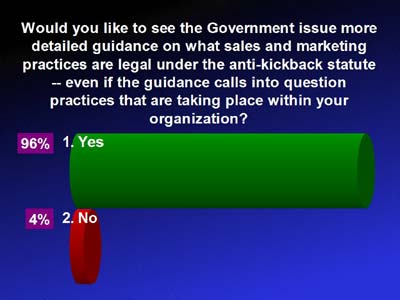
CMS PHARMACEUTICAL PRICE REPORTING GUIDANCE:
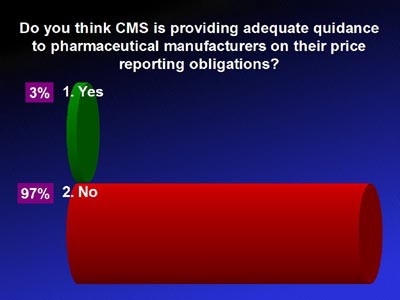
MERIDIA AUDIENCE RESPONSE:
These real time audience response results were generated by MERIDIA Audience Response, www.meridiaARS.com. MERIDIA is an interactive meeting consulting company which specializes in providing an audience with instant feedback throughout the conference.
CD-ROM OF ALL PRESENTATIONS AVAILABLE:
The Pharmaceutical Regulatory and Compliance Congress and Best Practices Forum is now offering a CD-ROM which includes video of all speakers plus synced Power Point presentations of the recent PharmaCongress, November 12-14, 2003 in Washington, DC. The CD-ROM costs only $295. Three or more CD-ROMs can be purchased for $225 each; Ten or more CD-ROMs cost $195 each. The CD-ROMs can be purchased at http://www.pharmacongress.com/cdrom/cdinfo4.html.
KEYNOTE SPEAKERS:
Thomas L. Abrams
Director, Division of Drug Marketing, Advertising & Communications, U.S. Food and Drug Administration, Rockville, MD
Arthur L. Caplan, Ph.D.
Director, Center for Bioethics, University of Pennsylvania, Fellow, American Association for the Advancement of Science, the Hastings Center, the College of Physicians of Philadelphia, and the New York Academy of Medicine, Philadelphia, PA
Dara Corrigan
Acting Principal Deputy Inspector General, Office of Inspector General, Department of Health and Human Services, Washington, DC
Raymond Gilmarti
Chairman, President and CEO, Merck & Co., Inc., Whitehouse Station, NJ
Thomas Grissom
Center for Medicare and Medicaid Services, Department of Health and Human Services, Washington, DC
John Guthrie, Esq.
Director, Medicaid Fraud Control Unit, Office of Attorney General, Columbus, OH
Thomas B. Leary, Esq.
Commissioner, Federal Trade Commission, Washington, DC
Dennis M. Nally
Chairman and Senior Partner, PricewaterhouseCoopers LLP, New York, NY
Janet Woodcock, MD
Director, Center for Drug Evaluation and Research, Food and Drug Administration, Rockville, MD
CONGRESS CO-CHAIRS:
Douglas M. Lankler, Esq.
Senior Corporate Counsel, Deputy Compliance Officer, Pfizer Inc., New York, NY
Arjun Rajaratnam, Esq.
Compliance Officer, Global Pharmaceuticals, GlaxoSmithKline, Research Triangle Park, NC
Bert Weinstein, Esq.
Vice President and Assistant General Counsel, Merck & Co., Whitehouse Station, NJ
SPONSOR: THE PHARMACEUTICAL COMPLIANCE FORUM:
The Congress is sponsored by the Pharmaceutical Compliance Forum (PCF). PCF was founded in early-1999 by compliance professionals from the pharmaceutical industry, to promote effective corporate compliance programs. The organization is open to others in the industry, and has more than doubled its membership since it was founded. The members meet twice a year, for 1-2 days, focusing on open and informal sharing of compliance information, best practices and current developments in the field.
CD-ROM ORDER INFORMATION:
For CD-ROM ordering Information, go to the Pharma Congress website: http://www.PharmaCongress.com.
For on-line purchase, visit:
http http://www.pharmacongress.com/cdrom/cdinfo4.html.
For more info, call 800-684-4549 or email: registration@hcconferences.com
Pharma Congress Office
7790 Barberry Ave.
Yucca Valley, CA USA 92284
Please forward this news announcement to your friends and colleagues who might find it useful.
FOR E-MAIL ADDRESS CHANGE, ADD OR DELETE REQUESTS:
For changes or additions, please email your request to: webmaster@PharmaCongress.com.
For removal of your e-mail address, please click the link below for "SafeUnsubscribe" to automatically remove your address from the list.
|